C. Jensen, H. Hohmeyer and G. Lysek; March 2002 (unpublished)
Summary
196 strains of Monilia fructigena (also called Sclerotinia fructigena (Pers.) Schröter) isolated for different types of rhythmic behavior also exhibited different ageing. About 80 % of the isolates grew linearly for 8 to 14 days before growth ceased (type L). The type „A“ (asterisk‑like) colonies grew likewise, then stopped but later distinct points at the hyphal front started again growing, the outbreaks produced asterisk-like shape of the colony. Type „S“ colonies grew slowly and stopped after days entirely. These types of senescence are compared to those known from Podospora pauciseta.
Introduction
It has been pointed out that the general concept of potential unlimited fungal growth cannot be maintained (GOTTLIEB, 1971). So far only a few filamentous fungi are known to stop their growth after a specific period of time without any external influence. In this aspect Podospora pauciseta (CES.) TRAV. (= Podospora anserina in lit.) is the most extensively studied object and the ageing habit of this pyrenomycete has been called „senescence“. It has been shown that each strain of P. pauciseta has a limited life span characterized by active growth, transformation to a senescent state, followed by cessation of growth. The course of senescence is characteristic for a given race (ESSER & TUDZINSKY, 1979). Likewise, the natural onset of senescence was observed in Rhizoctonia solani KÜHN and some other hyphomycetes (GOTTLIEB, 1971). Moreover, there is a comparable ageing behavior in the Aspergillus glaucus group called „vegetative death“. In contrast to the stopping growth the degenerative parts of the mycelium form sectors (CATEN & HANDLEY, 1978). Mutational induced cessation of growth was reported for Neurospora crassa SHEAR & DODGE, e.g. the stop-start phenotype in stopper and some poky mutants (McDOUGALL & PITTENGER, 1966).
In searching for the genetically fundamentals of ageing it became obvious that it is due to the presence and activity of any extra nuclear DNA, which can be infective by cytoplasmic fusion. The ability to transfer the senescent phenotype to a non-senescent mycelium via anastomosis as well as the maternal inheritance of the senescence determinants are the most striking evidences for a cytoplasmic basis of this phenomenon. In Podospora it has been shown that a mycelium passes through three physiological conditions (ESSER & TUDZINSKY, 1979):
(1) A non-senescent state, where the growth rate reaches its maximum and remains constant over a certain time.
(2) A pre-senescent state, where physiological and molecular genetically alterations occur to transfer the organism to the final senescent state. No morphological expressions are notable.
(3) A senescent state, where the growth rate progressively decreases. The peripheral hyphae swell and burst. This is followed by cellular death.
It will be demonstrated that Monilia fructigena (PERS.) PERS. resembles in many aspects the above described ageing habits.
Materials and methods
196 strains of Monilia fructigena were isolated from sporodochia gained from the surface of „ring rot“ infected apples, pears and plums collected in different sites in Berlin (West) (JENSEN & LYSEK, 1983). For isolation the conidia were inoculated to a malt-extract-apple juice agar. No attempt was made to obtain genetically homogenous material. The cultures were grown in light-dark-cycles (LD) or in continuous dark at 27 ° C (DD).

Results and discussion
The strains isolated represented three different growth patterns according to their type of senescence:
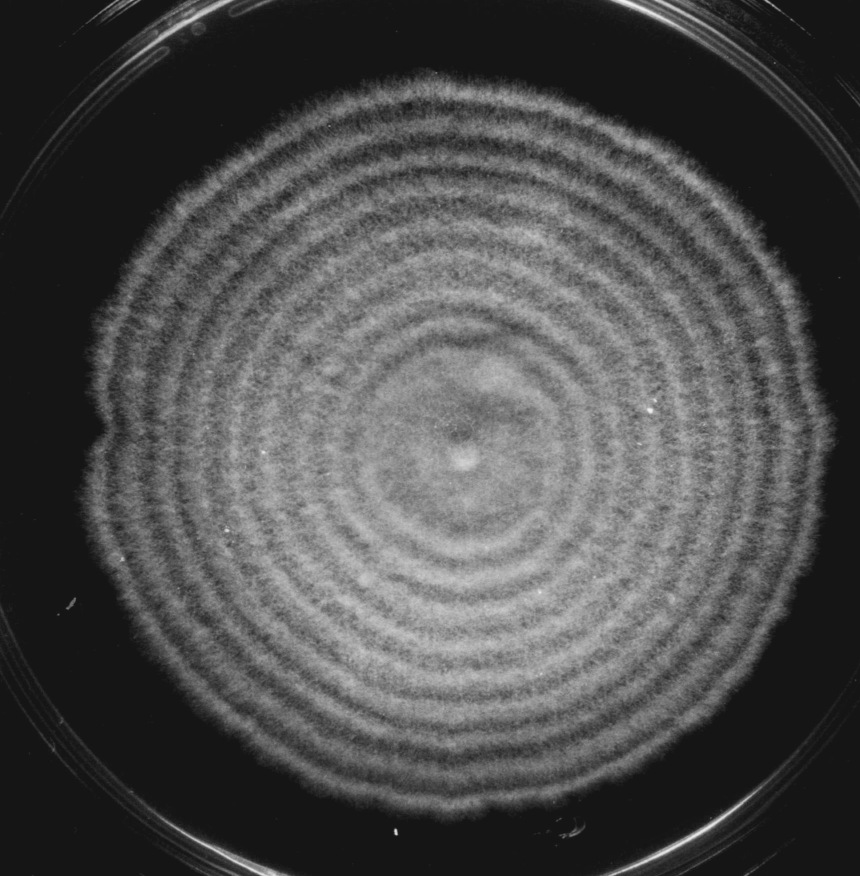
- Type „L“ („linear“ growth, fig. 1) showed up to 14 days a linear growth at a mean rate of 2,7 +/- 0,04 mm per day (for cultural conditions see JENSEN & LYSEK, 1983). This type was recognized by well coordinated zonations of the mycelium and a distinctly confined edge of the colony. After inoculation 3-4 days passed before zonations were formed. The linear growth continued about a week, seldom two weeks. Finally, the propagation of the mycelium decreased progressively and then stopped. Type L was observed in about 80 % of all cultures, whether they grew in LD or DD, and is said to be the normal form. The growth pattern agreed in principal aspects with the course of senescence in pauciseta, for instance. The main differences to the latter organism are so far:
(a) The senescence occurs earlier. (b) It was not observed that drastically morphological aberrations, e.g. „thinning“ of the mycelium in consequence of reduction of the aerial hyphae, took place before the growth ceased. (c) No lengthening of life span was observed while growing under continuous dark conditions. However, the mean diameter of a DD-culture was considerably reduced. (d) Four strains investigated intensively in order to keep an eye on their rhythmic behavior didn’t variate their linear growth type from 22°C to 32°C.
Frequently the colony of the L-type revealed sector-like gaps at the edge of a terminated colony. These were initiated by minor parts of the propagating mycelium, which stagnate a few days before the growth ceased, whereas the adjacent hyphae continue growing. In Aspergillus species the vegetative death syndrome displays as a first indication sectors of such a kind. On the other hand, the expression of either fast or slow growing sectors in M. fructigena isn’t a rare event.
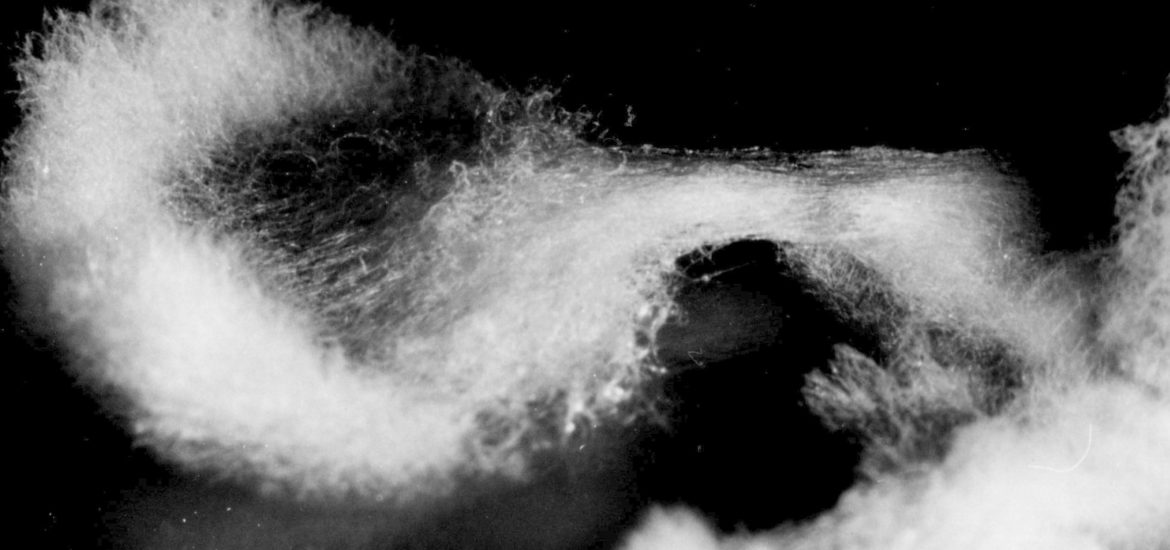
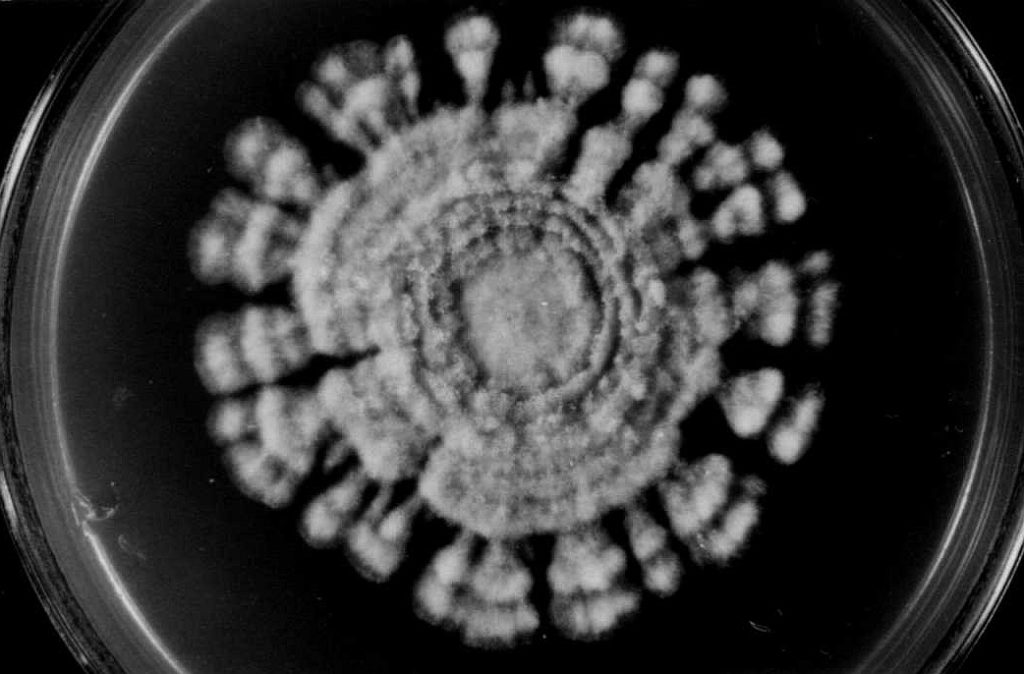
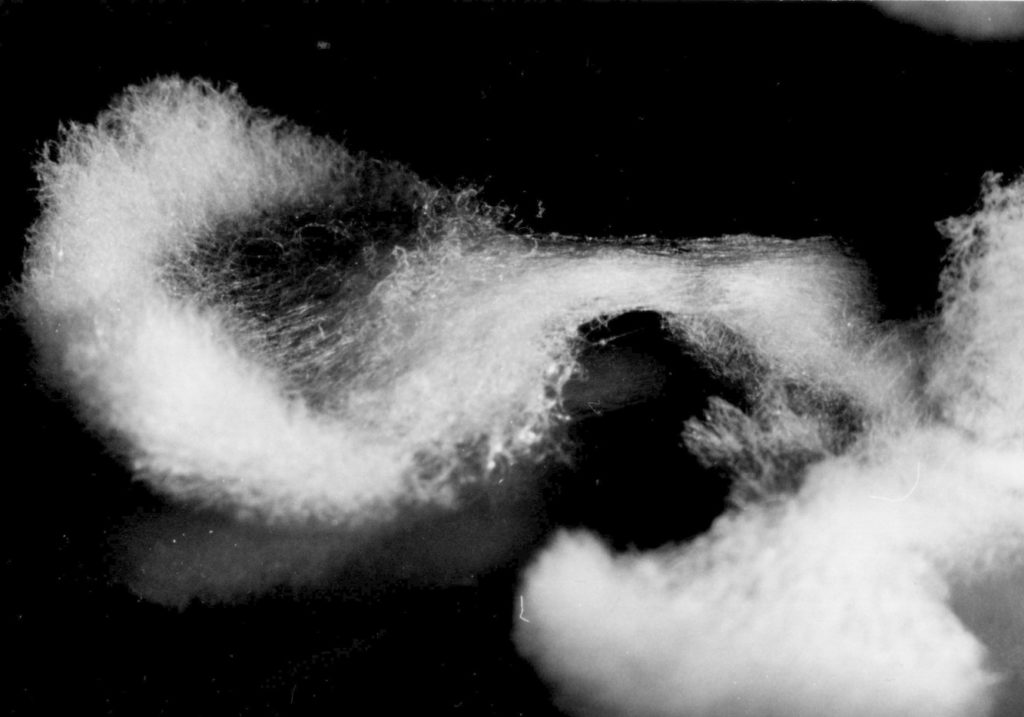
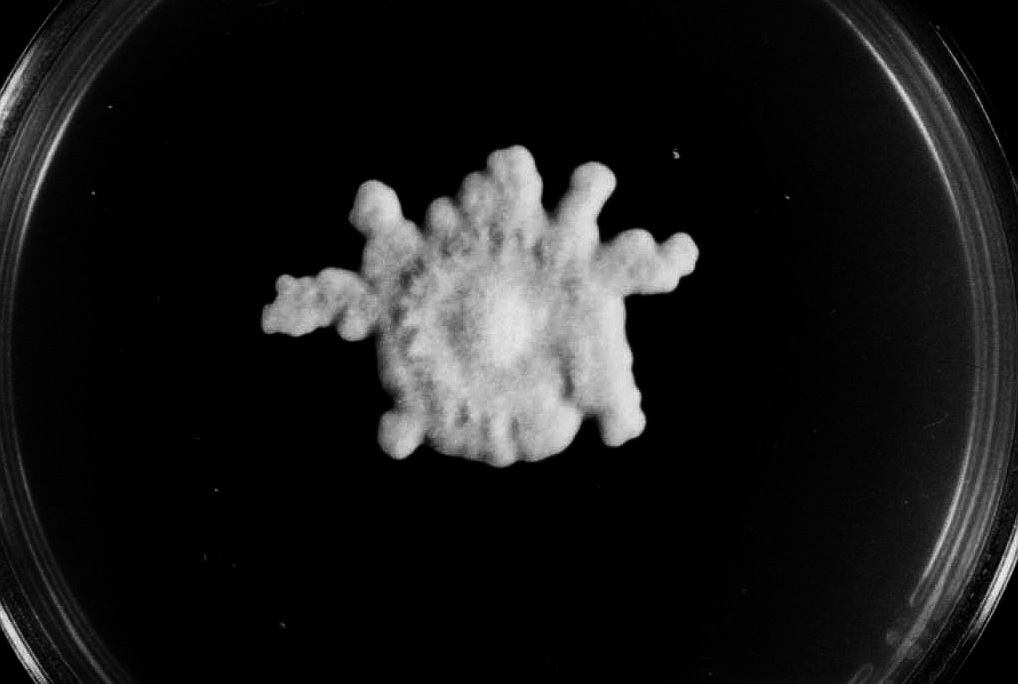
Considering the L-type the two following growth patterns are less abundant and said to be aberrations of the normal form: - In type „A“ („asterisk‑like“ growth, fig. 2 a) the growth rate was as fast as in type L or only slightly reduced. But after cessation and considerable pause of hyphal growth the colonies formed many sectors starting from precise points at the edge of the culture, so that it figures like an asterisk. The „out-breaks“ were growing rhythmically, as well. on malt extract agar medium a remarkable strategy to restart growing was observed: The aerial hyphae joined together forming dense strands. Then bridge-like structures overarched the edge of the colony and a small space of the medium to restart growth from the point they reached the medium (fig. 2b). Exceptionally the resumption of growth occurred continuously at the entire edge of the colony (fig. 2 c). Whatever strategy was taken, these patterns represent actually a stop-start-growth phenomenon similar to that in crassa. However, type A of M. fructigena differed in frequency, spontaneous and early occurrence as well as its different habits.
- Type „S“ („slow“ growth, fig. 3) showed a slow and usually non‑rhythmic growth, which after a few days stopped entirely. It is suggested that the type-S-cultures were isolated from already in their natural habitat predisposed sporodochia. First, this form may result from an inoculum bearing „presenescent“ material according to pauciseta, where subcultures from presenescent mycelia showed a reduced growth length. on the other hand, it might not be excluded that infection by fungal viruses or another external infective principle causes such a growth habit. However, without further investigations it cannot be decided, what the S-type determinates. Only about 8.5 % of the in LD conditions growing strains were of this type, but about 16 % of the DD-cultures.
Considering the diverse above described phenomena M. fructigena might be a new tool for further investigations in senescence and ageing. In this respect it has indeed some advantages: The organism occurs frequently in its natural habitat and is easy to isolate from its hosts. Senescence ensues always a relatively short period of continuous growth. The latter properties make this fungus easy to handle in laboratory cultures. There are any different morphological expressions of ageing, i.e. degenerative sectors, cessation of mycelium propagation and/or stop-start mode of growth. As the most critical disadvantage M. fructigena does not form a teleomorph in culture. Even in nature such an event is rarely observed. On the contrary sexual reproductive structures are important in senescence studies, since isolation and subcultivation of spores is the usual method to recover juvenile mycelia. Specific techniques are needed to overcome this problem.
Appendix: As amendment please see to pictures 4 and 5 which display the bridge building phenomenon.

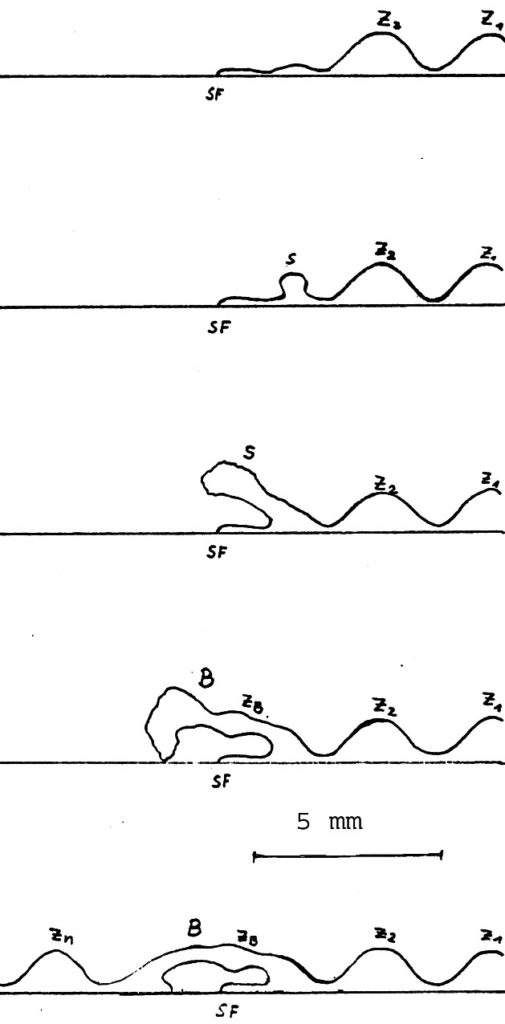
a) cessation and pause of hyphal growth, Z1 and Z2 last well formed zonations, SF = cessation front
b) beginning forming of aerial hyphae
c) aerial hyphae joined together forming dense strands (S)
d) bridge (B) meeting fresh agar surface
e) new zonations (Zn) after secondary starting point. Substrate hyphae not painted.
References
- CATEN, C.E. & HANDLEY, L. (1978). „Vegetative death“ Syndrome in Aspergillus glaucus. .Bulletin of the British Mycological Society 12, 114
- ESSER, K. & TUDZINSKY, P. (1979). Genetic control and expression of senescence in Podospora anserina. In Lemke, P.A. (ed.), Viruses and Plasmids in Fungi, N.Y.
- GOTTLIEB, D. (1971). Limited growth in Fungi. Mycologia 63, 619‑629
- JENSEN, C. & LYSEK G. (1983).Differences in the mycelial growth rhythms in a population of Sclerotinia fructigena. Experientia 39, 1401‑1402
- MCDOUGALL, K.J. & PITTENGER, T.H. (1966). A cytoplasmic variant of Neurospora crassa. Genetics 54, 551‑565.